|
EPITAXIAL DEPOSITION IN VAPORS AND SOLUTIONS In addition, the orientation of the crystal may deviate because of unintended nucleation by the container or stress caused by the difference in thermal expansion between the container and the crystal during growth. However, even though a seed crystal is used, single crystal metals produced by the Bridgman method are typically of lower quality than those produced by the Czochralski method because the melt and the growing crystal are in contact with the container so that the container material may be included as an impurity on the edge of the crystal. A better-quality single crystal can be obtained when a seed crystal is used. 9 The most ideal solution for the supercooling and nucleation problems is the use of a single crystal seed. These problems can be overcome by remelting some of the nucleated crystals or by controlling the mold design (a conical end to generate a nucleus at one point or a small channel to allow only one nucleus having a fast growing crystallographic orientation to pass into the melt). 7 In the Bridgman method, because nuclei are generated by supercooling of the melt at the liquid-solid interface, the crystal growth rate from these nuclei in the initial stage is very fast, resulting in the formation of a single crystal with many defects or a polycrystal in the presence of multiple nuclei. 7,8 By adjusting the shape of the container or mold, the shape of the single crystal product can be controlled. A container or a mold filled with the metal is passed through a furnace, which imposes a temperature gradient to produce a single crystal from the melt by unidirectional solidification from one end to the other. In addition, the heat transport is more easily controlled by using a furnace rather than the complex rotation and pulling processes as in the Czochralski method. The Bridgman method is simpler than the Czochralski method because it is not based on mass transport, only heat transport. These surface properties are especially important for the heteroepitaxial growth of other materials such as thin films and in catalysis for various chemical reactions at the surface. 3 Single crystal metals generally have a uniform surface orientation and relatively flat surfaces compared to polycrystalline metals. The scattering of electrons at GBs is temperature dependent because it is related to electron-phonon coupling, but it has been found that at temperatures below about 70 K, such scattering does not contribute significantly to the change in electrical conductivity because the motion of phonons is suppressed at this temperature. 2 GBs in polycrystalline metals scatter electrons so that single crystal metals have a higher electrical conductivity at room temperature. 
Single crystal metals with extremely large grains are generally weaker and more ductile than polycrystalline metals. This limits the plasticity and hence increases the yield strength of metals. During deformation, dislocations propagate in the grain through the appropriate slip systems and GBs impede their motion so that they “pile up” at GBs. 1 The mechanics of single crystal metals differ from polycrystalline metals because the strength of a metal is strongly related to the size of the grain. 
For example, they have anisotropic properties (e.g., elastic constants, linear thermal expansion, linear compressibility, and electrical resistance), while randomly oriented polycrystalline metals usually have isotropic properties. 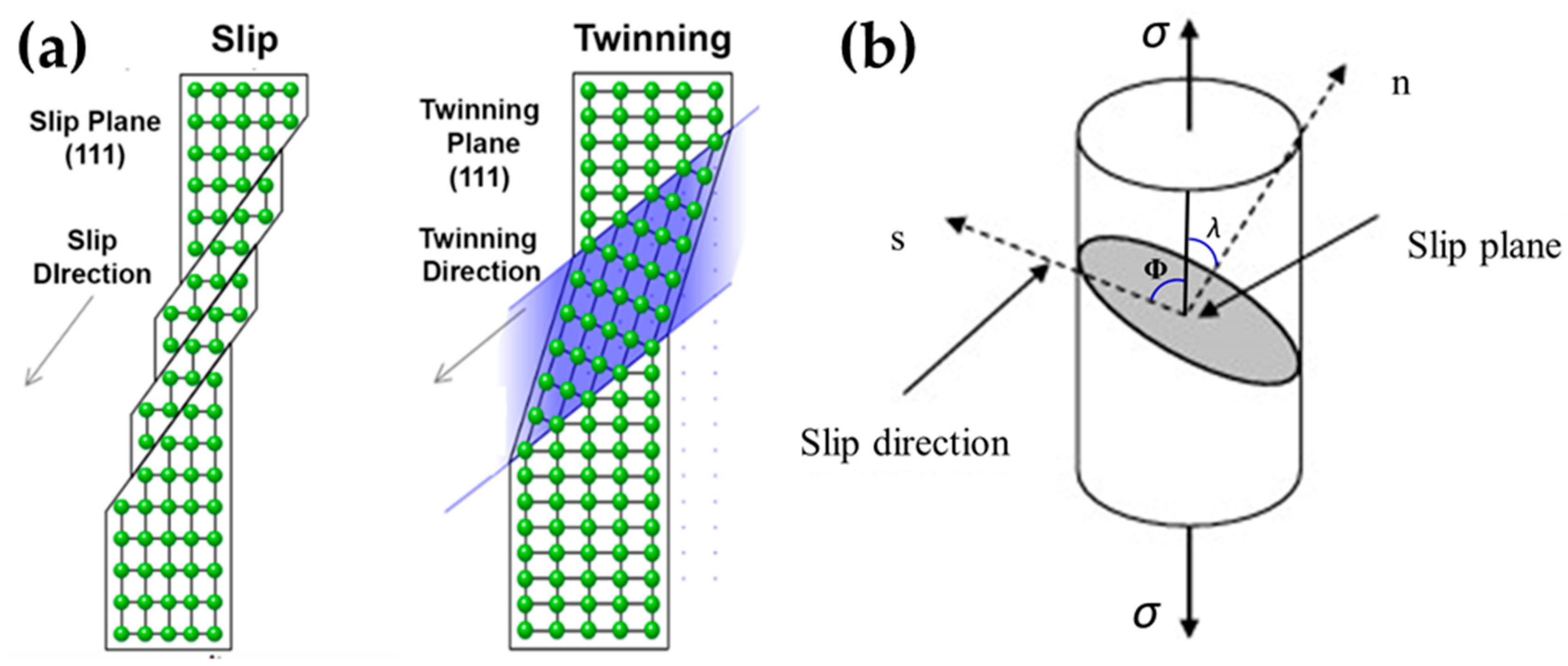
Single crystal metals have different properties and uses than their polycrystalline counterparts. Most metals in general use are polycrystalline, thus consisting of grains separated by grain boundaries (GBs), whereas single crystal metals have a single grain throughout the entire sample and have no GBs. A grain in metallurgy is a small or microscopic single crystal, which has a periodic arrangement of atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
